First Principles Calculation of Electronic Structure of Spinel Manganese Oxide Doping with Transition Metal
-
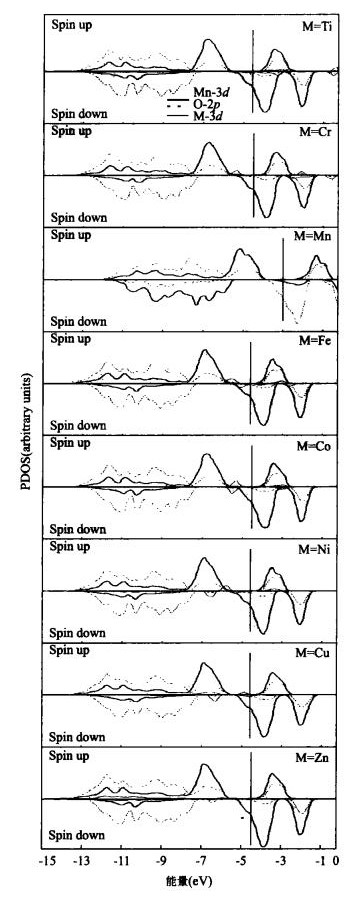
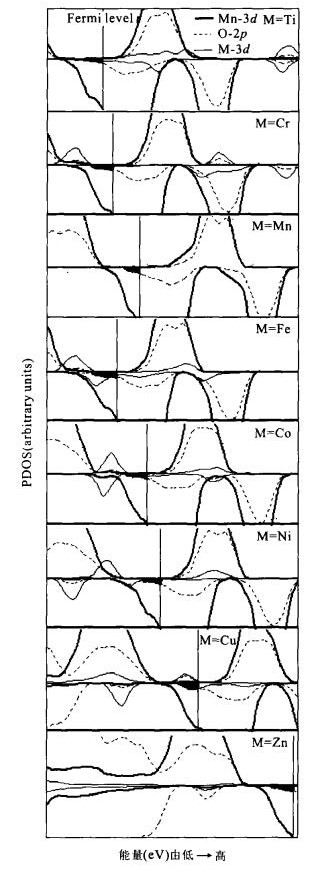
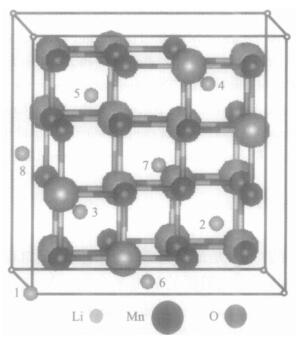
摘要: 尽管对过渡金属掺杂锰酸锂后放电平台的升高现象有众多实验研究, 但对其机理的研究却鲜见报道.采用第一原理的密度泛函理论, 计算了过渡金属M(M=Ti、Cr、Fe、Co、Ni、Cu、Zn)掺杂尖晶石型LiMn2O4的电子结构, 并以此分析放电平台的升高机理.电子态密度分析发现由于M-3d能带的诱导作用, 出现了新的O-2p能带, 而锂脱出时获得的电子, 主要是由费米能级附近O-2p能带提供的.当过渡金属M由Ti变化到Zn时, M-3d能带逐渐向低能量的方向移动, 新的O-2p能带出现的位置也随之下移, 当Li脱出时, 需要更多的能量才能从低能量的O-2p能带上获得电子, 因而体系能够获得较高的嵌入电压.
-
关键词:
- 尖晶石型LIMN2O4 /
- 电子结构 /
- 第一原理 /
- 掺杂
Abstract: Although there has been intensive research on improving the discharge voltage plateau of lithium manganese oxide doped by transition metals, there is a lack of corresponding studies on the improvement mechanism. In this paper, we investigate the electronic structure of spinel manganese oxide doping with transition metal M (M=Ti, Cr, Fe, Co, Ni, Cu, Zn) by first principles calculation based on the density functional theory. The calculated density of states indicates that a new O-2p band induced by the M-3d band appears at the exact position of the M-3d band itself. The compensating electrons are removed from the O-2p levels neighboring the Fermi level when the Li ion is removed. The position of the M-3d band shifts gradually to the low energy direction as M varies from Ti to Zn in the transition metal row of the periodic table, and the position of the new O-2p band also shifts downward following the M-3d band. The withdrawal of lithium electrons from this low O-2p band will result in high cell voltage.-
Key words:
- spinel manganese oxide /
- electronic structure /
- first principles /
- doping
-
-
Artacho, E., Sánchez-Portal, D., Ordejón, P., et al., 1999. Linear-scaling ab-initio calculations for large and complex systems. Phys. Status Solidi. B, 215: 809-817. doi: 10.1002/(SICI)1521-3951(199909)215:1<809::AID-PSSB809>3.0.CO;2-0 Aydinol, M.K., Kohn, A.F., Ceder, G., et al., 1997. Ab initio study of lithium intercalation in metal oxides and metal dichalcogenides. Phys. Rev. B, 56: 1354-1365. doi: 10.1103/PhysRevB.56.1354 Ceder, G., Aydinol, M.K., Kohn, A.F., 1997. Application of first-principles calculations to the design of rechargeable Li-batteries. Comput. Mater. Sci., 8: 161-169. doi: 10.1016/S0927-0256(97)00029-3 He, X.M., Li, J.J., Cai, Y., et al., 2005. Preparation of codoped spherical spinel LiMn2O4cathode materials for Liion batteries. Journal of Power Sources, 150: 216-222. doi: 10.1016/j.jpowsour.2005.02.029 Liu, H.X., Zhou, Z.P., Zhao, S.X., 2001. The microwave synthesis of LiMn2O4 electron materials. Journal of Physical Chemistry, 17: 702-707(in Chinese with English abstract). Markovskya, B., Talyossef, Y., Salitra, G., et al., 2004. Cycling and storage performance at elevated temperatures of LiNi0.5Mn1.5O4 positive electrodes for advanced 5 V Li-ion batteries. Electrochemistry Communications, 6: 821-826. doi: 10.1016/j.elecom.2004.06.005 Ning, L.C., Wu, J.P., Zhou, C.G., et al., 2006. On the influence of sequential lithium insertions on the physical properties of spinel manganese oxide. International Journal of Quantum Chemistry(in Press). Park, S.H., Sun, Y.K., 2004. Synthesis and electrochemical properties of 5 V spinel LiNi0.5Mn1.5O4cathode materials prepared by ultrasonic spray pyrolysis method. Electrochimica Acta, 50: 429-432. Shi, S. Q., Ouyang, C. Y., Wang, D.S., et al., 2003. The effect of cation doping on spinel LiMn2O4: A firstprinciples investigation. Solid State Communications, 126: 531-534. doi: 10.1016/S0038-1098(03)00234-5 Song, G.M., Li, W.J., Zhou, Y., 2004. Synthesis of Mg-doped LiMn2O4powders for lithium-ion batteries by rotary heating. Materials Chemistry and Physics, 87: 162-167. doi: 10.1016/j.matchemphys.2004.05.023 Tasnádi, F., Nagy, Á., 2002. Local self-interaction-free approximate exchange-correlation potentials in the variational density functional theory for individual excited states. Chemical Physics Letters, 366: 496-503. doi: 10.1016/S0009-2614(02)01612-3 Xia, Y. Y., Yoshio, M., 1997. Studies on Li-Mn-O spinel system(obtained from melt-impregnation method)as a cathode for 4 V lithium batteries, Part Ⅳ. High and low temperature performance of LiMn2O4. Journal of Power Sources, 66: 129-133. 刘韩星, 周振平, 赵世玺, 2001. LiMn2O4体系电极材料的微波合成. 物理化学学报, 17: 702-707. doi: 10.3866/PKU.WHXB20010807 -








 下载:
下载: