Experimental Petrology: Status Quo and Prospect
-
摘要: 实验岩石学通过高温高压实验来模拟地球内部状态,正演研究矿物、岩石及其组分的物理化学行为,与天然矿物和岩石样品反演研究相互补充. 从20世纪初美国卡内基研究所建立地球物理实验室算起,实验岩石学已经历了100多年的发展,在认识地球内部状态和过程以及矿物和岩石成因方面发挥了重要作用. 我国实验岩石学研究开展约50年,进入21世纪以来在实验平台和创新性研究成果方面取得了显著进步. 在学科发展趋势方面,实验岩石学表现出以下6方面的特点:(1)新的高温高压实验技术不断涌现;(2)实验与分析测试技术高度融合;(3)实验模拟与计算模拟相结合;(4)从热力学平衡扩展到动力学研究;(5)从干体系扩展到对挥发分和流体的深入研究;(6)应用场景从固体地球扩展到类地行星. 通过进一步开发或改进高温高压实验技术,加强与分析测试技术以及计算技术的结合,实验岩石学有望在破解地球内部流体的性质和作用、地幔演化和岩浆分异、变质反应速率和机制、类地行星形成与演化等重要科学问题方面作出关键贡献.Abstract: Through simulating the high pressure and high temperature conditions in Earth's interior, experimental petrology investigates the physicochemical behavior of minerals, rocks and the components they contain, which complements "inverse problem" study using natural samples. Since the foundation of the Geophysical Laboratory of the Carnegie institution in the early 20th century, the development of experimental petrology has been taking place for more than 100 years. Experimental petrology has played a crucial role in advancing our knowledge about the conditions and processes in Earth's interior and the genesis of minerals and rocks. In China, experimental petrology has developed for more than five decades, and significant progress has been made in the 21th century with respect to laboratory building and scientific research. We highlight the following characteristics in the development of experimental petrology as a discipline: (1) emergence of novel high temperature and high pressure experimental techniques; (2) integration of experimental and analytical techniques; (3) combination of experimental simulation and computational simulation; (4) expansion from thermodynamic equilibrium to kinetics; (5) expansion from dry systems to volatiles-bearing systems including fluids; (6) expansion from the Earth to other terrestrial planets. Through further development in experimental techniques and more intimate combination with analytical and computational methods, experimental petrology is expected to make important contributions in resolving important scientific problems, such as the properties and effects of geofluids, mantle evolution and magma differentiation, rates and mechanisms of metamorphic reactions, and the formation and evolution of terrestrial planets.
-
Key words:
- experimental petrology /
- high temperature /
- high pressure /
- in situ measurement
-
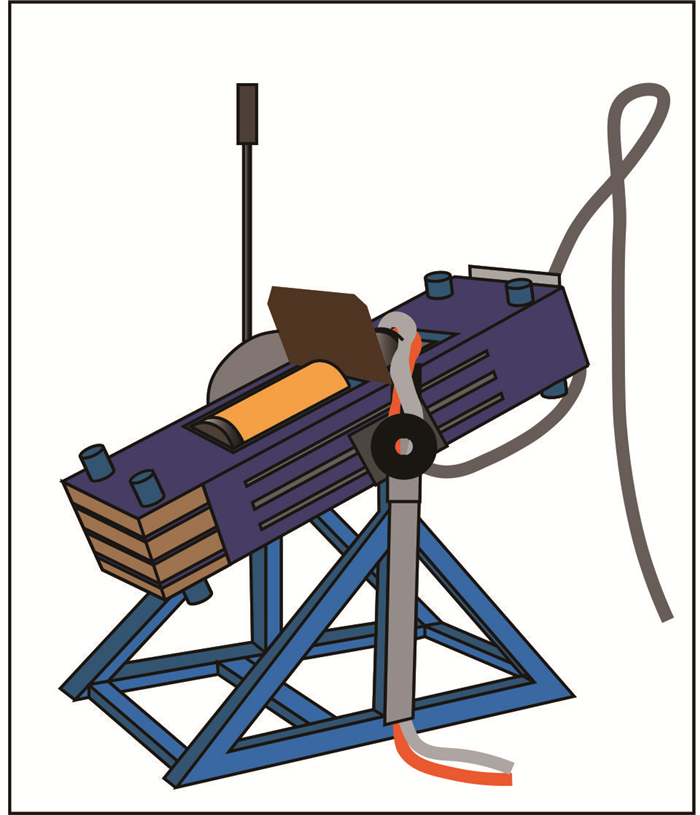
图 1 瑞士ETH研制的摇摆式多面砧压机
改自Schmidt and Ulmer(2004)
Fig. 1. Rocking multianvil device developed by ETH Zürich, Switzerland
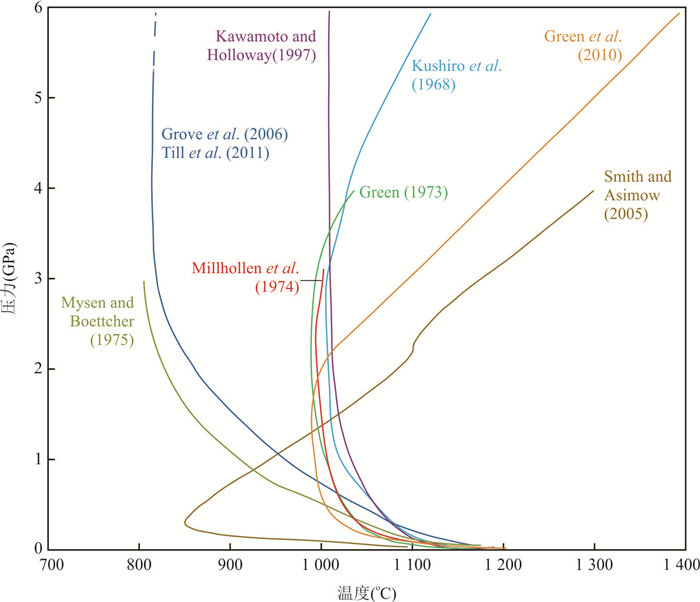
图 3 关于地幔橄榄岩湿固相线的巨大争议
Fig. 3. Discrepancy in the H2O‐saturated solidus of mantle peridotite
-
Ardia, P., Giordano, D., Schmidt, M. W, 2008. A Model for the Viscosity of Rhyolite as a Function of H2O‐Content and Pressure: a Calibration Based on Centrifuge Piston Cylinder Experiments. Geochimica et Cosmochimica Acta, 72(24): 6103-6123. https://doi.org/10.1016/j.gca.2008.08.025 Bassett, W. A., Shen, A. H., Bucknum, M., et al., 1993. A New Diamond Anvil Cell for Hydrothermal Studies to 2.5 GPa and from -190 to 1 200 ℃. Review of Scientific Instruments, 64(8): 2340-2345. https://doi.org/10.1063/1.1143931 Belonoshko, A. B., Saxena, S. K, 1992. A Unified Equation of State for Fluids of C‐H‐O‐N‐S‐Ar Composition and Their Mixtures up to very High Temperatures and Pressures. Geochimica et Cosmochimica Acta, 56(10): 3611-3626. https://doi.org/10.1016/0016‐7037(92)90157‐E Bowen, N. L., 1928. The Evolution of the Igneous Rocks. Princeton University Press, Princeton. Boyd, F. R., England, J. L, 1960. Apparatus for Phase‐Equilibrium Measurements at Pressures up to 50 Kilobars and Temperatures up to 1 750 ℃. Journal of Geophysical Research, 65(2): 741-748. https://doi.org/10.1029/jz065i002p00741 Cashman, K. V., Sparks, R. S. J., Blundy, J. D, 2017. Vertically Extensive and Unstable Magmatic Systems: a Unified View of Igneous Processes. Science, 355(6331): eaag3055. https://doi.org/10.1126/science.aag3055 Chen, W., Xiong, X. L., Wang, J. T., et al., 2018. TiO2 Solubility and Nb and Ta Partitioning in Rutile‐Silica‐Rich Supercritical Fluid Systems: Implications for Subduction Zone Processes. Journal of Geophysical Research: Solid Earth, 123(6): 4765-4782. Costa, F., 2021. Clocks in Magmatic Rocks. Annual Review of Earth and Planetary Sciences, 49: 231-252. https://doi.org/10.1146/annurev‐earth‐080320‐060708 Duan, Z. H., Zhang, Z. G., 2006. Equation of State of the H2O, CO2, and H2O‐CO2 Systems up to 10 GPa and 2 573.15 K: Molecular Dynamics Simulations with Ab Initio Potential Surface. Geochimica et Cosmochimica Acta, 70(9): 2311-2324. https://doi.org/10.1016/j.gca.2006.02.009 Fichtner, C. E., Schmidt, M. W., Liebske, C., et al., 2021. Carbon Partitioning between Metal and Silicate Melts during Earth Accretion. Earth and Planetary Science Letters, 554: 116659. https://doi.org/10.1016/j.epsl.2020.116659 Forman, R. A., Piermarini, G. J., Barnett, J. D., et al., 1972. Pressure Measurement Made by the Utilization of Ruby Sharp‐Line Luminescence. Science, 176(4032): 284-285. https://doi.org/10.1126/science.176.4032.284 Ghiorso, M. S., Sack, R. O., 1995. Chemical Mass Transfer in Magmatic Processes Ⅳ. a Revised and Internally Consistent Thermodynamic Model for the Interpolation and Extrapolation of Liquid‐Solid Equilibria in Magmatic Systems at Elevated Temperatures and Pressures. Contributions to Mineralogy and Petrology, 119(2/3): 197-212. https://doi.org/10.1007/BF00307281 Green, D. H., Hibberson, W. O., Rosenthal, A., et al., 2014. Experimental Study of the Influence of Water on Melting and Phase Assemblages in the Upper Mantle. Journal of Petrology, 55(10): 2067-2096. https://doi.org/10.1093/petrology/egu050 Green, D.H., 1973. Experimental Melting Studies on a Model Upper Mantle Composition at High‐Pressure under Water‐Saturated and Water‐Undersaturated Conditions. Earth and Planetary Science Letters, . 19: 37-53. https://doi.org/10.1016/0012‐821X(73)90091‐5 Green, D.H., Hibberson, W.O., Kovacs, I., Rosenthal, A., 2010. Water and Its Influence on the Lithosphere‐Asthenosphere Boundary. Nature, 467(7314): 448-451. https://doi.org/10.1038/nature09369 Grove, T., Till, C., Krawczynski, M., 2012. The Role of H2O in Subduction Zone Magmatism. Annual Review of Earth and, 40(1): 413-439. doi: 10.1146/annurev‐earth‐042711‐105310 Grove, T.L., Chatterjee, N., Parman, S.W., Medard, E., 2006. The Influence of H2O on Mantle Wedge Melting. Earth and Planetary Science Letters, . 249(1): 74-89. https://doi.org/10.1016/j.epsl.2006.06.043 Guo, X., Zhang, L., Behrens, H., et al., 2016. Probing the Status of Felsic Magma Reservoirs: Constraints from the P‐T‐H2O Dependences of Electrical Conductivity of Rhyolitic Melt. Earth and Planetary Science Letters, 433: 54-62. https://doi.org/10.1016/j.epsl.2015.10.036 Holland, T. J. B., Powell, R., 1990. An Enlarged and Updated Internally Consistent Thermodynamic Dataset with Uncertainties and Correlations: The System K2O‐Na2O‐CaO‐MgO‐MnO‐FeO‐Fe2O3‐Al2O3‐TiO2‐SiO2‐C‐H2‐O2. Journal of Metamorphic Geology, 8(1): 89-124. Holland, T. J. B., Powell, R., 2011. An Improved and Extended Internally Consistent Thermodynamic Dataset for Phases of Petrological Interest, Involving a New Equation of State for Solids. Journal of Metamorphic Geology, 29(3): 333-383. https://doi.org/10.1111/j.1525‐1314.2010.00923.x Jamtveit, B., 2010. Metamorphism: From Patterns to Processes. Elements, 6(3): 149-152. https://doi.org/10.2113/gselements.6.3.149 Jiang, D. W., Gao, Y., Cao, M., et al., 2021. Diamond Anvil Cell with Double Coaxial Chambers. Review of Scientific Instruments, 92(12): 123901. https://doi.org/10.1063/5.0063573 Karki, B. B., 2015. First‐Principles Computation of Mantle Materials in Crystalline and Amorphous Phases. Physics of the Earth and Planetary Interiors, 240: 43-69. https://doi.org/10.1016/j.pepi.2014.11.004 Kawai, N., 1966. A Static High Pressure Apparatus with Tapering Multi‐Pistons Forming a Sphere. I. Proceedings of the Japan Academy, 42(4): 385-388. https://doi.org/10.2183/pjab1945.42.385 Kawamoto, T., Holloway,J.R., 1997. Melting Temperature and Partial Melt Chemistry of H2O‐Saturated Mantle Peridotite to 11 Gigapascals. Science, 276(5310): 240-243. https://doi.org/10.1126/science.276.5310.240 Kirkpatrick, R.J., 1975. Crystal Growth from the Melt: a Review. American Mineralogist, 60: 798-814. Kushiro, I., Syono, Y., Akimoto, S., 1968. Melting of a Peridotite Nodule at High Pressures and High Water Pressures. Journal of Geophysical Research, 73(18): 6023-6029. https://doi.org/10.1029/JB073i018p06023 Li, J. K., Bassett, W. A., Chou, I. M., et al., 2016. An Improved Hydrothermal Diamond Anvil Cell. Review of Scientific Instruments, 87(5): 053108. https://doi.org/10.1063/1.4947506 Liu, L. P., Hu, X. M., Liu, X., 2018. MgO Partition between Olivine and K2O‐Rich Silicate Melt: Geothermometers Applicable to High Potassium Magmas. Journal of Asian Earth Sciences, 166: 181-194. https://doi.org/10.1016/j.jseaes.2018.07.036 Liu, X., Zhang, L. F., Alistair, H., et al., 2009. Effect of Water on the Partial Melting Process of some Silicate Systems: Important Implication of the Second Critical Endpoint. Acta Petrologica Sinica, 25(12): 3407-3421(in Chinese with English abstract). Luth, W.C., Tuttle, O.F., 1963. Externally Heated Cold‐Seal Pressure Vessels for Use to 10 000 Bars and 750℃. American Mineralogist, 48: 1401-1403. Mibe, K., Kawamoto, T., Matsukage, K. N., et al., 2011. Slab Melting Versus Slab Dehydration in Subduction‐Zone Magmatism. Proceedings of the National Academy of Sciences of the United States of America, 108(20): 8177-8182. https://doi.org/10.1073/pnas.1010968108 Millhollen, G.L., Irving, A.J., Wyllie, P.J., 1974. Melting Interval of Peridotite with 5.7% Water to 30 Kilobars. The Journal of Geology, 82(5): 575-587. https://doi.org/10.2307/30059149 Murakami, M., Hirose, K., Kawamura, K., et al., 2004. Post‐Perovskite Phase Transition in MgSiO3. Science, 304(5672): 855-858. https://doi.org/10.1126/science.1095932 Mysen, B.O., Boettcher, A.L., 1975. Melting of a Hydrous Mantle: Ⅰ. Phase Relations of Natural Peridotite at High‐Pressures and Temperatures with Controlled Activities of Water, Carbon Dioxide, and Hydrogen. Journal of Petrology, 16(1): 520-548. https://doi.org/10.1093/petrology/16.1.520 Ni, H. W., 2015. Oxygen Isotope Thermometry, Speedometry, and Hygrometry: Apparent Equilibrium Temperature Versus Closure Temperature. Geochemistry, Geophysics, Geosystems, 16(1): 27-39. https://doi.org/10.1002/2014gc005574 Ni, H. W., Keppler, H., Walte, N., et al., 2014. In Situ Observation of Crystal Growth in a Basalt Melt and the Development of Crystal Size Distribution in Igneous Rocks. Contributions to Mineralogy and Petrology, 167(5): 1003. https://doi.org/10.1007/s00410‐014‐1003‐9. Ni, H. W., Zhang, L., Xiong, X. L., et al., 2017. Supercritical Fluids at Subduction Zones: Evidence, Formation Condition, and Physicochemical Properties. Earth‐Science Reviews, 167: 62-71. https://doi.org/10.1016/j.earscirev.2017.02.006 Ni, H. W., 2020. Properties and Effects of Supercritical Geofluids. Bulletin of Mineralogy, Petrology and Geochemistry, 39(3): 443-447 (in Chinese with English abstract). Oganov, A. R., Ono, S., 2004. Theoretical and Experimental Evidence for a Post‐Perovskite Phase of MgSiO3 in Earth Layer. Nature, 430(6998): 445-448. https://doi.org/10.1038/nature02701 Poe, B. T., McMillan, P. F., Rubie, D. C., et al., 1997. Silicon and Oxygen Self‐Diffusivities in Silicate Liquids Measured to 15 Gigapascals and 2 800 Kelvin. Science, 276(5316): 1245-1248. https://doi.org/10.1126/science.276.5316.1245 Schmidt, M. W., Poli, S., 2014. Devolatilization during Subduction. Treatise on Geochemistry. Elsevier, Amsterdam, 669-701. https://doi.org/10.1016/b978‐0‐08‐095975‐7.00321‐1 Schmidt, M. W., Ulmer, P., 2004. A Rocking Multianvil: Elimination of Chemical Segregation in Fluid‐Saturated High‐Pressure Experiments. Geochimica et Cosmochimica Acta, 68(8): 1889-1899. https://doi.org/10.1016/j.gca.2003.10.031. Sekine, T., Wyllie, P. J., 1983. Experimental Simulation of Mantle Hybridization in Subduction Zones. The Journal of Geology, 91(5): 511-528. https://doi.org/10.1086/628802 Shen, A. H., Keppler, H., 1997. Direct Observation of Complete Miscibility in the Albite‐H2O System. Nature, 385(6618): 710-712. https://doi.org/10.1038/385710a0 Smith, P.M., Asimow, P.D., 2005. Adiabat_1ph: a New Public Front‐End to the MELTS, pMELTS, and pHMELTS Models. Geochemistry, Geophysics, Geosystems, 6(2): Q02004. https://doi.org/10.1029/2004GC000816 Testemale, D., Argoud, R., Geaymond, O., et al., 2005. High Pressure/High Temperature Cell for X‐Ray Absorption and Scattering Techniques. Review of Scientific Instruments, 76(4): 043905. https://doi.org/10.1063/1.1884188 Till, C.B., Grove, T.L., Withers, A.C., 2011. The Beginnings of Hydrous Mantle Wedge Melting. Contributions to Mineralogy and Petrology, 163: 669-688. https://doi.org/10.1007/s00410‐011‐0692‐6 Wang, C. G., Liang, Y., Xu, W. L., et al., 2013. Effect of Melt Composition on Basalt and Peridotite Interaction: Laboratory Dissolution Experiments with Applications to Mineral Compositional Variations in Mantle Xenoliths from the North China Craton. Contributions to Mineralogy and Petrology, 166(5): 1469-1488. https://doi.org/10.1007/s00410‐013‐0938‐6 Wang, Q. X., Zhou, D. Y., Li, W. C., et al., 2021. Spinodal Decomposition of Supercritical Fluid Forms Melt Network in a Silicate‐H2O System. Geochemical Perspectives Letters, 22-26. https://doi.org/10.7185/geochemlet.2119 Watson, E. B., 1981. Diffusion in Magmas at Depth in the Earth: The Effects of Pressure and Dissolved H2O. Earth and Planetary Science Letters, 52(2): 291-301. https://doi.org/10.1016/0012‐821X(81)90184‐9 Weir, C. E., Lippincott, E. R., Van Valkenburg, A., et al., 1959. Infrared Studies in the 1‐ to 15‐Micron Region to 30, 000 Atmospheres. Journal of Research of the National Bureau of Standards Section A, Physics and Chemistry, 63A(1): 55-62. https://doi.org/10.6028/jres.063A.003 Xie, H.S., 1997. An Introduction to Material Science of Deep Earth. Science Press, Beijing (in Chinese). Xu, J., Mao, H., 2000. Moissanite: a Window for High‐Pressure Experiments. Science, 290(5492): 783-785. https://doi.org/10.1126/science.290.5492.783 Yang, X. Z., Liu, D. D., Xia, Q. K., 2014. CO2‐Induced Small Water Solubility in Olivine and Implications for Properties of the Shallow Mantle. Earth and Planetary Science Letters, 403: 37-47. https://doi.org/10.1016/j.epsl. 2014. 06.025 doi: 10.1016/j.epsl.2014.06.025 Yoder, H. S. Jr., 1950. High‐Low Quartz Inversion up to 10, 000 Bars. Transactions, American Geophysical Union, 31(6): 827. https://doi.org/10.1029/tr031i006p00827 Zhang, J. F., Wang, C., Wang, Y. F., 2012. Experimental Constraints on the Destruction Mechanism of the North China Craton. Lithos, 149: 91-99. https://doi.org/10.1016/j.lithos.2012.03.015 Zhang, L., Guo, X., Wang, Q. X., et al., 2017. Diffusion of Hydrous Species in Model Basaltic Melt. Geochimica et Cosmochimica Acta, 215: 377-386. https://doi.org/10.1016/j.gca.2017.07.019 Zhang, Y. X., Cherniak, D. J., 2010. Diffusion in Minerals and Melts: Introduction. Reviews in Mineralogy and Geochemistry, 72: 1-4. Zhang, Y. X., Jenkins, J., Xu, Z. J., 1997. Kinetics of the Reaction H2O+O→2OH in Rhyolitic Glasses Upon Cooling: Geospeedometry and Comparison with Glass Transition. Geochimica et Cosmochimica Acta, 61(11): 2167-2173. https://doi.org/10.1016/S0016‐7037(97)00054‐9 Zhang, Y. X., Stolper, E. M., Ihinger, P. D., 1995. Kinetics of the Reaction H2O+O=2OH in Rhyolitic and Albitic Glasses; Preliminary Results. American Mineralogist, 80(5/6): 593-612. https://doi.org/10.2138/am‐1995‐5‐618 刘曦, 张立飞, Hack, C.A., 等, 2009. 水对硅酸盐体系部分熔融行为的影响: 第二临界端点的重要意义. 岩石学报, 25(12): 3407-3421. https://www.cnki.com.cn/Article/CJFDTOTAL-YSXB200912028.htm 倪怀玮, 2020. 超临界地质流体的性质和效应. 矿物岩石地球化学通报, 39(3): 443-447, 440. https://www.cnki.com.cn/Article/CJFDTOTAL-KYDH202003003.htm 谢鸿森, 1997. 地球深部物质科学导论. 北京: 科学出版社. -








 下载:
下载: