The Morphological Distribution and Geochemical Genesis of Antimony in Typical High-Temperature Hot Springs
-
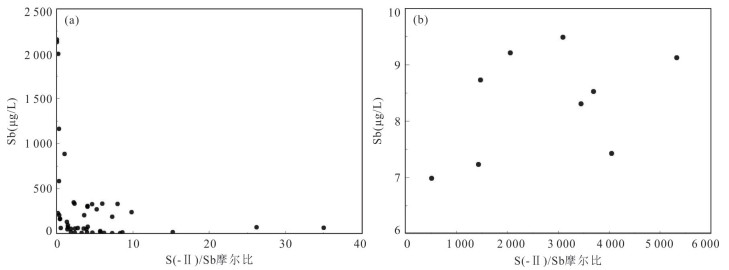
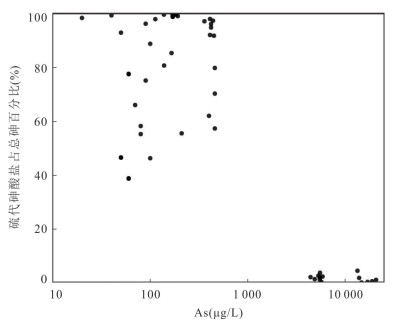
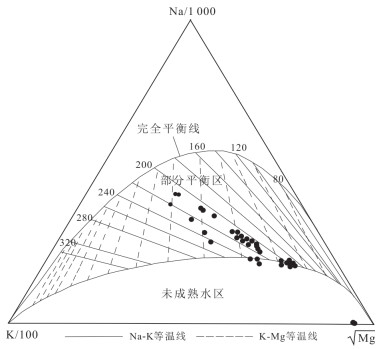
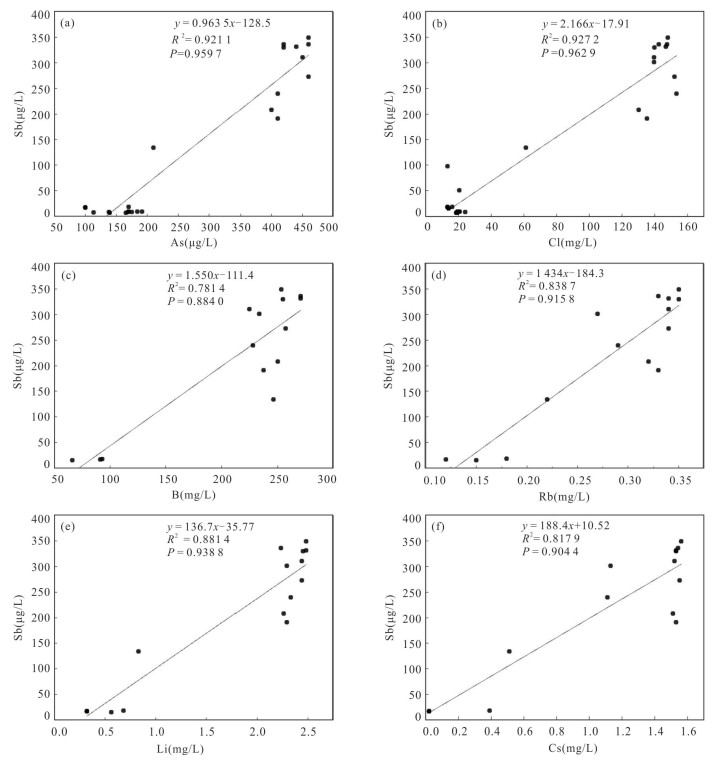
摘要: 锑为典型有害元素,地热成因锑是天然水环境中溶解态锑的重要来源,富锑热泉排泄的负面环境效应不容忽视.本文在藏南和滇西选择典型地热区,分析了热泉中锑的形态分布及其地球化学成因.总体上,研究区排泄的地热水具有远高于天然水环境背景值的锑含量,最高可达2 128.7 μg/L.水文地球化学计算表明热泉中锑的主要存在形态为锑酸盐和亚锑酸盐,但部分热泉样品中硫代锑占总锑百分比可高达35%.硫化物浓度、S(-Ⅱ)/Sb摩尔比,以及砷锑之间的竞争巯基化作用是影响热泉中硫代锑含量的关键因素.在本研究所涉及地热系统中,西藏玛旁雍错曲色涌巴、门士、莫落江为岩浆热源型地热系统,其地热水中锑源自高温条件下热储围岩淋滤和作为热源的岩浆房所释出流体的输入,西藏曲卓木、朗久与云南邦腊掌则属于非岩浆热源型地热系统,其热泉中的锑主要来源于地热水‒围岩矿物相互作用.Abstract: Antimony is a typical harmful element, and the negative environmental effects of antimony-rich hot springs discharge can not be neglected since geothermal genesis of antimony is an important source of antimony in the natural water environments. In this paper, the morphological distribution of antimony in hot springs and its geochemical genesis were analyzed in selected typical geothermal areas in southern Tibet and western Yunnan. In general, the antimony of geothermal water discharged from the study area can be up to 2 128.7 μg/L, which is much higher than the background values in the natural water environments. Hydrogeochemical calculations indicate that the main forms of antimony present in the hot springs are antimonate and antimonite, however, the percentage of thioantimony in some hot spring samples can be as high as 35% of the total antimony. Sulfide concentration, S(-Ⅱ)/Sb molar ratio, and competitive thiolation between arsenic and antimony are the key factors affecting the content of thioantimony in hot springs. Among the geothermal systems involved in this study, the Tibetan MapamYumco, Moincer, and Moluojiang are magma-heat geothermal systems, and the antimony in the geothermal water originates from the input of fluids released from the hot storage surrounding rocks leaching and magma house as the heat source under high temperature conditions, while the Tibetan Quzuomu, Langjiu and Yunnan Banglazhang are non-magma-heat geothermal systems, and the main source of antimony in their thermal springs is geothermal water-peripheral rock mineral interaction.
-
Key words:
- hot springs /
- antimony /
- thioantimony /
- water‐rock interactions /
- magmatic fluid input /
- geochemistry
-
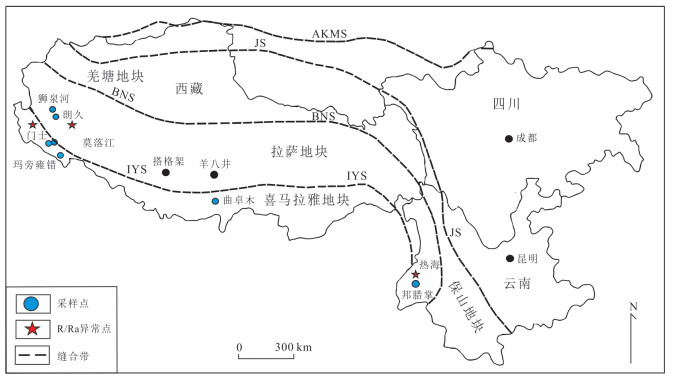
图 1 采样区域分布(据Guo et al., 2019修改)
Fig. 1. Distribution of sampling areas (modified from Guo et al., 2019)
表 1 热泉水文地球化学特征
Table 1. Hydrogeochemical characteristics of hot springs
样品编号 地热区 T (℃) pH EC (μs/cm) Sb (μg/L) H2S (mg/L) As (μg/L) LJ01 朗久 78.0 7.84 2 992 78.9 0.09 5 510 LJ02 朗久 65.0 8.36 3 037 65.1 0.01 5 690 LJ03 朗久 69.9 8.08 2 926 72.5 0.03 5 480 LJ04 朗久 63.0 7.94 3 239 57.0 0.03 4 860 LJ05 朗久 84.4 8.13 3 259 64.7 0.05 5 510 LJ06 朗久 82.0 8.41 3 116 60.6 0.06 5 890 LJ07 朗久 83.2 8.34 3 142 55.1 0.06 5 310 QSYB01 玛旁雍错曲色涌巴 85.0 8.51 1 715 301.3 0.34 360 QSYB02 玛旁雍错曲色涌巴 88.4 8.53 1 766 336.1 0.22 460 QSYB03 玛旁雍错曲色涌巴 84.0 8.32 2 077 240.1 0.66 410 QSYB04 玛旁雍错曲色涌巴 76.0 9.20 1 760 207.8 0.21 400 QSYB05 玛旁雍错曲色涌巴 75.0 8.94 1 818 191.2 0.39 410 QSYB06 玛旁雍错曲色涌巴 70.0 8.92 1 848 310.6 0.35 450 QSYB07 玛旁雍错曲色涌巴 70.0 8.90 1 828 330.2 0.43 420 QSYB08 玛旁雍错曲色涌巴 66.0 8.96 1 804 335.7 0.56 420 QSYB09 玛旁雍错曲色涌巴 65.0 8.96 1 818 331.1 0.74 440 QSYB10 玛旁雍错曲色涌巴 90.0 9.13 1 845 349.3 0.22 460 QSYB11 玛旁雍错曲色涌巴 84.0 9.09 1 796 272.7 0.40 460 QSYB12 玛旁雍错曲色涌巴 84.0 2.40 3 014 134.2 0.05 210 MS01 门士 71.2 7.50 1 482 18.8 0.08 170 MS01-1 门士 69.0 8.05 1 414 18.7 0.03 90 MS01-2 门士 67.5 7.88 1 491 18.1 0.02 100 MS01-3 门士 64.1 8.01 1 460 16.6 0.04 100 MS01-4 门士 60.5 7.95 1 431 15.3 0.02 20 MS02 门士 71.7 7.69 1 461 50.5 0.02 70 MS03 门士 72.5 7.72 1 452 98.0 0.04 90 MLJ01 莫落江 83.3 7.30 6 453 1 164.5 0.10 20 750 MLJ02 莫落江 82.0 7.80 5 590 2 157.6 0.03 17 030 MLJ03 莫落江 79.0 8.21 6 225 584.1 0.05 18 940 MLJ04 莫落江 87.0 8.33 5 870 2 128.7 0.01 14 770 MLJ05 莫落江 87.0 8.48 5 447 1 996.2 0.11 13 940 MLJ06 莫落江 85.0 8.19 5 468 885.5 0.26 13 390 QZM01 曲卓木 70.0 6.95 1 995 168.6 0.02 40.0 QZM02 曲卓木 65.0 6.53 1 670 8.60 0.02 50.0 QZM03 曲卓木 69.0 7.16 1 970 163.9 0.02 80.0 QZM04 曲卓木 72.0 6.87 2 037 11.5 0.02 60.0 QZM05 曲卓木 67.0 7.17 2 057 207.5 0.02 60.0 QZM06 曲卓木 62.0 6.98 2 085 19.2 0.01 50.0 QZM07 曲卓木 58.0 7.29 2 082 230.1 0.01 50.0 QZM08 曲卓木 54.0 7.60 2 054 15.2 0.01 60.0 QZM09 曲卓木 74.0 6.73 2 101 10.9 0.01 60.0 QZM10 曲卓木 71.0 8.26 1 071 9.82 0.02 80.0 LL00 邦腊掌 91.0 8.36 1 080 8.31 8.00 168.8 LL01 邦腊掌 75.0 7.97 1 054 9.22 5.30 183.8 LL02 邦腊掌 87.0 8.45 1 045 8.53 8.80 174.9 LL03 邦腊掌 88.0 9.13 1 075 9.49 8.20 191.4 LL04 邦腊掌 63.0 8.25 1 000 6.99 1.00 139.0 LL05 邦腊掌 84.5 8.58 1 053 8.73 3.60 138.2 LL06 邦腊掌 57.5 8.73 938 7.43 8.40 112.8 LL07 邦腊掌 94.0 9.23 1 057 9.13 13.60 170.0 LL08 邦腊掌 76.0 9.67 981 7.23 2.90 165.4 表 2 水中不同锑形态的化学热力学数据
Table 2. Chemical thermodynamic data of various tungsten species in water
名称 化学式 反应式 平衡
常数焓变
(kJ/mol)数据来源 Tetrathioantimonate Sb2S42‒ 2Sb(OH)3+4HS‒+2H+=Sb2S42‒+6H2O 49.39 ‒321.78 University of Waterloo-
minteqv4uw.datAntimonate SbO3‒ Sb(OH)6‒=SbO3‒+3H2O 2.93 -- SbO2+ Sb(OH)6‒+2H+=SbO2++4H2O 2.39 -- Sb(OH)6‒ Sb2O5+7H2O=2Sb(OH)6‒+2H+ ‒27.82 -- Antimonite Sb(OH)3 Sb(OH)6‒+2e‒+3H+=Sb(OH)3+3H2O 24.31 -- Sb(OH)4‒ Sb(OH)3+H2O=Sb(OH)4‒+H+ 12.04 69.85 Sb(OH)2+ Sb(OH)3+H+=Sb(OH)2++H2O 1.39 -- HSbO2 Sb(OH)3=HSbO2+H2O ‒0.01 ‒0.13 SbO2‒ Sb(OH)3=SbO2‒+H2O+H+ ‒11.80 70.186 6 SbO+ Sb(OH)3+H+=SbO++2H2O 0.92 8.242 5 SbOF Sb(OH)3+F‒+H+=SbOF+2H2O 6.19 -- Sb(OH)2F Sb(OH)3+F‒+H+=Sb(OH)2F+H2O 6.19 -- 表 3 热泉样品中锑的形态分布模拟结果
Table 3. Simulation results of the morphological distribution of antimony in hot spring samples
样品编号 Sb Sb(Ⅲ) Sb(Ⅴ) Sb2S42- (mol/L) (mol/L) (%) (mol/L) (%) (mol/L) (%) LJ01 6.52E-07 6.48E-07 99.76 1.57E-09 0.24 9.22E-29 0 LJ02 5.38E-07 6.22E-08 11.61 4.74E-07 88.39 5.32E-32 0 LJ03 5.99E-07 1.24E-07 20.79 4.73E-07 79.21 1.20E-30 0 LJ04 4.71E-07 1.42E-07 30.19 3.28E-07 69.81 1.14E-29 0 LJ05 5.35E-07 9.41E-08 17.67 4.38E-07 82.33 8.32E-32 0 LJ06 5.01E-07 4.12E-08 8.26 4.58E-07 91.74 1.98E-31 0 LJ07 4.55E-07 4.72E-08 10.41 4.06E-07 89.59 1.97E-31 0 QSYB01 2.49E-06 2.48E-06 99.99 1.46E-10 0.01 5.17E-15 0 QSYB02 2.78E-06 2.77E-06 100 1.69E-11 0 1.31E-17 0 QSYB03 1.98E-06 1.98E-06 100 1.07E-11 0 1.96E-12 0 QSYB04 1.72E-06 1.71E-06 99.78 3.75E-09 0.22 1.07E-18 0 QSYB05 1.58E-06 1.57E-06 99.91 1.41E-09 0.09 1.28E-15 0 QSYB06 2.57E-06 2.55E-06 99.93 1.69E-09 0.07 1.60E-14 0 QSYB07 2.73E-06 2.71E-06 99.92 2.17E-09 0.08 1.19E-13 0 QSYB08 2.77E-06 2.76E-06 99.87 3.69E-09 0.13 1.89E-12 0 QSYB09 2.74E-06 2.72E-06 99.85 4.00E-09 0.15 1.28E-11 0 QSYB10 2.89E-06 2.87E-06 99.96 1.19E-09 0.04 1.21E-20 0 QSYB11 2.25E-06 2.24E-06 100 2.81E-11 0 7.76E-18 0 QSYB12 1.11E-06 1.10E-06 100 1.19E-11 0 4.71E-32 0 MS01 1.55E-07 1.54E-07 99.88 1.89E-10 0.12 3.83E-16 0 MS01-1 1.55E-07 3.76E-08 24.43 1.16E-07 75.57 3.17E-22 0 MS01-2 1.49E-07 5.12E-08 34.37 9.77E-08 65.63 2.70E-24 0 MS01-3 1.37E-07 3.45E-08 25.27 1.02E-07 74.73 4.88E-20 0 MS01-4 1.27E-07 4.38E-08 34.80 8.20E-08 65.20 8.75E-16 0 MS02 4.18E-07 2.08E-07 50.07 2.07E-07 49.93 1.35E-22 0 MS03 8.10E-07 3.74E-07 46.36 4.32E-07 53.64 1.24E-17 0 MLJ01 9.62E-06 7.78E-06 80.91 1.83E-06 19.09 1.68E-30 0 MLJ02 1.78E-05 7.48E-06 42.05 1.03E-05 57.95 4.28E-31 0 MLJ03 4.83E-06 5.05E-07 10.47 4.31E-06 89.53 1.82E-31 0 MLJ04 1.76E-05 2.35E-06 13.40 1.52E-05 86.60 2.27E-33 0 MLJ05 1.65E-05 9.92E-07 6.03 1.54E-05 93.97 1.27E-29 0 MLJ06 7.32E-06 9.47E-07 12.98 6.35E-06 87.02 2.61E-28 0 MLJ07 4.91E-07 2.19E-07 44.75 2.70E-07 55.25 7.08E-29 0 MLJ08 2.59E-07 4.75E-08 18.35 2.11E-07 81.65 6.73E-33 0 QZM01 1.39E-06 1.22E-06 88.24 1.63E-07 11.76 1.72E-14 0 QZM02 7.07E-08 6.83E-08 96.50 2.47E-09 3.50 9.10E-22 0 QZM03
QZM041.35E-06
9.46E-081.08E-06
8.55E-0880.16
90.332.68E-07
9.14E-0919.84
9.671.62E-22
3.65E-240
0QZM05 1.71E-06 1.37E-06 80.35 3.35E-07 19.65 2.04E-20 0 QZM06 1.58E-07 1.37E-07 86.90 2.07E-08 13.10 1.58E-24 0 QZM07 1.89E-06 1.42E-06 74.87 4.76E-07 25.13 2.75E-21 0 QZM08 1.25E-07 6.86E-08 54.84 5.65E-08 45.16 2.91E-23 0 QZM09 8.97E-08 8.37E-08 93.38 5.94E-09 6.62 1.03E-27 0 QZM10 8.08E-08 1.04E-08 12.88 7.04E-08 87.12 1.08E-24 0 LL00 6.83E-08 6.83E-08 99.98 6.34E-13 0 7.34E-12 0.01 LL01 7.58E-08 2.83E-08 37.38 8.39E-16 0 2.37E-08 31.15 LL02 7.01E-08 7.01E-08 99.94 5.45E-13 0 1.99E-11 0.03 LL03 7.80E-08 7.78E-08 99.75 1.99E-10 0.25 9.51E-16 0 LL04 5.74E-08 5.60E-08 97.48 8.22E-15 0 7.23E-10 1.25 LL05 7.17E-08 7.17E-08 100 7.02E-13 0 2.39E-13 0 LL06 6.11E-08 1.80E-08 29.40 5.99E-15 0 2.16E-08 35.12 LL07 7.50E-08 7.38E-08 98.30 1.28E-09 1.70 9.06E-17 0 LL08 5.94E-08 5.88E-08 98.94 6.32E-10 1.06 8.91E-19 0 注:E-07即10-7,依此类推. -
Aksoy, N., Şimşek, C., Gunduz, O., 2009. Groundwater Contamination Mechanism in a Geothermal Field: A Case Study of Balcova, Turkey. Journal of Contaminant Hydrology, 103(1-2): 13-28. https://doi.org/10.1016/j.jconhyd.2008.08.006 Boreiko, C. J., Rossman, T. G., 2020. Antimony and Its Compounds: Health Impacts Related to Pulmonary Toxicity, Cancer, and Genotoxicity. Toxicology and Applied Pharmacology, 403: 115156. https://doi.org/10.1016/j.taap.2020.115156 Cavallo, D., Iavicoli, I., Setini, A., et al., 2002. Genotoxic Risk and Oxidative DNA Damage in Workers Exposed to Antimony Trioxide. Environmental and Molecular Mutagenesis, 40(3): 184-189. https://doi.org/10.1002/em.10102 Dong, S. L., Zhang, Z., Zhang, L. K., et al., 2018. Geochemistry, Hf-Sr-Nd Isotopes and Petrogenesis of Acidic Volcanic Rocks in Quzhuomu Region of Southern Tibet. Earth Science, 43(8): 2701-2714 (in Chinese with English abstract). Filella, M., Belzile, N., Chen, Y. W., 2002. Antimony in the Environment: A Review Focused on Natural Waters. Earth-Science Reviews, 59(1-4): 265-285. https://doi.org/10.1016/S0012-8252(02)00089-2 Giggenbach, W. F., 1988. Geothermal Solute Equilibria. Derivation of Na-K-Mg-Ca Geoindicators. Geochimica et Cosmochimica Acta, 52(12): 2749-2765. https://doi.org/10.1016/0016-7037(88)90143-3 Giggenbach, W. F., Soto, R. C., 1992. Isotopic and Chemical Composition of Water and Steam Discharges from Volcanic-Magmatic-Hydrothermal Systems of the Guanacaste Geothermal Province, Costa Rica. Applied Geochemistry, 7(4): 309-332. https://doi.org/10.1016/0883-2927(92)90022-U Guo, Q. H., 2020. Magma-Heated Geothermal Systems and Hydrogeochemical Evidence of Their Occurrence. Acta Geologica Sinica, 94(12): 3544-3554 (in Chinese with English abstract). doi: 10.3969/j.issn.0001-5717.2020.12.002 Guo, Q. H., Liu, M. L., Li, J. X., et al., 2017a. Fluid Geochemical Constraints on the Heat Source and Reservoir Temperature of the Banglazhang Hydrothermal System, Yunnan-Tibet Geothermal Province, China. Journal of Geochemical Exploration, 172: 109-119. https://doi.org/10.1016/j.gexplo.2016.10.012 Guo, Q. H., Planer-Friedrich, B., Liu, M. L., et al., 2017b. Arsenic and Thioarsenic Species in the Hot Springs of the Rehai Magmatic Geothermal System, Tengchong Volcanic Region, China. Chemical Geology, 453: 12-20. https://doi.org/10.1016/j.chemgeo.2017.02.010 Guo, Q. H., Planer-Friedrich, B., Liu, M. L., et al., 2019. Magmatic Fluid Input Explaining the Geochemical Anomaly of very High Arsenic in some Southern Tibetan Geothermal Waters. Chemical Geology, 513: 32-43. https://doi.org/10.1016/j.chemgeo.2019.03.008 Guo, Q. H., Yang, C., 2021. Tungsten Anomaly of the High-Temperature Hot Springs in the Daggyai Hydrothermal Area, Tibet, China. Earth Science, 46(7): 2544-2554 (in Chinese with English abstract). He, M. C., Wan, H. Y., 2004. Distribution, Speciation, Toxicity and Bioavailability of Antimony in the Environment. Progress in Chemistry, 16(1): 131-135 (in Chinese with English abstract). doi: 10.3321/j.issn:1005-281X.2004.01.020 Hoke, L., Lamb, S., Hilton, D. R., et al., 2000. Southern Limit of Mantle-Derived Geothermal Helium Emissions in Tibet: Implications for Lithospheric Structure. Earth and Planetary Science Letters, 180(3-4): 297-308. https://doi.org/10.1016/S0012-821X(00)00174-6 Hou, Z. Q., Li, Z. Q., 2004. Possible Location for Underthrusting Front of the Indus Continent: Constraints from Helium Isotope of the Geothermal Gas in Southern Tibet and Eastern Tibet. Acta Geologica Sinica, 78(4): 482-493 (in Chinese with English abstract). doi: 10.3321/j.issn:0001-5717.2004.04.007 Liao, Z. L., Liao, G. Y., Pan, G. T., et al., 2005. Distribution and Utilization of Geothermal Resources in Tibet Ngari. China Mining Magazine, 14(8): 43-46 (in Chinese with English abstract). doi: 10.3969/j.issn.1004-4051.2005.08.014 Lima, A., Cicchella, D., Di Francia, S., 2003. Natural Contribution of Harmful Elements in Thermal Groundwaters of Ischia Island (Southern Italy). Environmental Geology, 43(8): 930-940. https://doi.org/10.1007/s00254-002-0715-8 Liu, M. L., Guo, Q. H., Wu, G., et al., 2019. Boron Geochemistry of the Geothermal Waters from Two Typical Hydrothermal Systems in Southern Tibet (China): Daggyai and Quzhuomu. Geothermics, 82: 190-202. https://doi.org/10.1016/j.geothermics.2019.06.009 Liu, M. L., Cao, Y. W., Wang, M. D., et al., 2014. Source of Hydrochemical Composition and Formation Mechanism of Rehai Geothermal Water in Tengchong. Safety and Environmental Engineering, 21(6): 1-7 (in Chinese with English abstract). doi: 10.3969/j.issn.1671-1556.2014.06.001 Lou, H., Wang, C. Y., Huangfu, G., et al., 2002. Three-Demensional Seismic Velocity Tomography of the Upper Crust in Tengchong Volcanic Area, Yunnan Province. Acta Seismologica Sinica, 24(3): 243-251 (in Chinese with English abstract). doi: 10.3321/j.issn:0253-3782.2002.03.003 McCleskey, R. B., Nordstrom, D. K., Susong, D. D., et al., 2010. Source and Fate of Inorganic Solutes in the Gibbon River, Yellowstone National Park, Wyoming, USA. Journal of Volcanology and Geothermal Research, 193(3/4): 189-202. https://doi.org/10.1016/j.jvolgeores.2010.03.014 Planer-Friedrich, B., Wilson, N., 2012. The Stability of Tetrathioantimonate in the Presence of Oxygen, Light, High Temperature and Arsenic. Chemical Geology, 322-323: 1-10. https://doi.org/10.1016/j.chemgeo.2012.06.010 Poon, R., Chu, I., Lecavalier, P., et al., 1998. Effects of Antimony on Rats Following 90-Day Exposure via Drinking Water. Food and Chemical Toxicology, 36(1): 21-35. https://doi.org/10.1016/S0278-6915(97)80120-2 Shen, L. C., Wu, K. Y., Xiao, Q., et al., 2011. Study on CO2 Degassing in Geothermal Anomaly Areas in Tibet: A Case Study of Langjiu and Tagejia Geothermal Areas. Chinese Science Bulletin, 56(26): 2198-2208 (in Chinese). doi: 10.1360/csb2011-56-26-2198 Smichowski, P., 2008. Antimony in the Environment as a Global Pollutant: A Review on Analytical Methodologies for Its Determination in Atmospheric Aerosols. Talanta, 75(1): 2-14. https://doi.org/10.1016/j.talanta.2007.11.005 Wang, X. W., Wang, T. H., Gao, N. A., et al., 2022. Formation Mechanism and Development Potential of Geothermal Resources along the Sichuan-Tibet Railway. Earth Science, 47(3): 995-1011 (in Chinese with English abstract). Wang, Y. Y., Zeng, L. S., Hou, K. J., et al., 2022. Mantle Source Components and Magmatic Evolution for the Comei Large Igneous Province: Evidence from the Early Cretaceous Niangzhong Mafic Magmatism in Tethyan Himalaya. Journal of Earth Science, 33(1): 133-149. https://doi.org/10.1007/s12583-021-1464-5 Wilson, N., Webster-Brown, J., Brown, K., 2012. The Behaviour of Antimony Released from Surface Geothermal Features in New Zealand. Journal of Volcanology and Geothermal Research, 247-248: 158-167. https://doi.org/10.1016/j.jvolgeores.2012.08.009 Yan, K. T., Guo, Q. H., Liu, M. L., 2019. Geochemical Anomalies of Arsenic and Its Speciation in Daggyai Geothermal Springs, Tibet. Journal of Jilin University (Earth Science Edition), 49(2): 548-558 (in Chinese with English abstract). Zhao, C. P., 2008. Mantle-Derived Helium Release Characteristics and Deep Magma Chamber Activities of Present Day in the Tengchong Volcanic Area (Dissertation). Institute of Geology, China Earthquake Administration, Beijing (in Chinese with English abstract). 董随亮, 张志, 张林奎, 等, 2018. 藏南曲卓木地区酸性火山岩地球化学、Hf-Sr-Nd同位素特征及其成因. 地球科学, 43(8): 2701-2714. doi: 10.3799/dqkx.2018.165 郭清海, 2020. 岩浆热源型地热系统及其水文地球化学判据. 地质学报, 94(12): 3544-3554. doi: 10.3969/j.issn.0001-5717.2020.12.002 郭清海, 杨晨, 2021. 西藏搭格架高温热泉中钨的水文地球化学异常. 地球科学, 46(7): 2544-2554. doi: 10.3799/dqkx.2020.287 何孟常, 万红艳, 2004. 环境中锑的分布、存在形态及毒性和生物有效性. 化学进展, 16(1): 131-135. doi: 10.3321/j.issn:1005-281X.2004.01.020 侯增谦, 李振清, 2004. 印度大陆俯冲前缘的可能位置: 来自藏南和藏东活动热泉气体He同位素约束. 地质学报, 78(4): 482-493. doi: 10.3321/j.issn:0001-5717.2004.04.007 廖忠礼, 廖光宇, 潘桂棠, 等, 2005. 西藏阿里地热资源的分布特点及开发利用. 中国矿业, 14(8): 43-46. doi: 10.3969/j.issn.1004-4051.2005.08.014 刘明亮, 曹耀武, 王敏黛, 等, 2014. 腾冲热海热泉水化学组分来源及其形成机制探讨. 安全与环境工程, 21(6): 1-7. https://www.cnki.com.cn/Article/CJFDTOTAL-KTAQ201406001.htm 楼海, 王椿镛, 皇甫岗, 等, 2002. 云南腾冲火山区上部地壳三维地震速度层析成像. 地震学报, 24(3): 243-251. https://www.cnki.com.cn/Article/CJFDTOTAL-DZXB200203003.htm 沈立成, 伍坤宇, 肖琼, 等, 2011. 西藏地热异常区CO2脱气研究: 以朗久和搭格架地热区为例. 科学通报, 56(26): 2198-2208. https://www.cnki.com.cn/Article/CJFDTOTAL-KXTB201126009.htm 汪新伟, 王婷灏, 高楠安, 等. 2022. 川藏铁路沿线地热资源形成机理与开发潜力. 地球科学, 47(3): 995-1011. doi: 10.3799/dqkx.2022.059 严克涛, 郭清海, 刘明亮, 2019. 西藏搭格架高温热泉中砷的地球化学异常及其存在形态. 吉林大学学报(地球科学版), 49(2): 548-558. https://www.cnki.com.cn/Article/CJFDTOTAL-CCDZ201902021.htm 赵慈平, 2008. 腾冲火山区现代幔源氦释放特征及深部岩浆活动研究(博士学位论文). 北京: 中国地震局地质研究所. -








 下载:
下载: